Norwegian register for devices and EUDAMED
Published:
Changes
Norwegian manufacturers, assemblers, and authorized representatives are legally obligated to register themselves and their devices.
Registration contributes to the Norwegian Medical Products Agency's guidance for manufacturers, market surveillance, and the handling of reported incidents involving medical devices.
Registration Requirements for Medical Device Stakeholders
The registration requirement does not apply to foreign manufacturers and authorized representatives who place devices on the Norwegian market through a Norwegian importer or distributor. Distributors and importers also have no registration obligation.
| Actor | Norwegian register | Eudamed |
|---|---|---|
| Norwegian Manufacturer | Mandatory | Voluntary |
| In-house Manufacturer | No obligation | Eudamed not open for In-house manufacturers |
| Norwegian Authorised representative | Mandatory | Voluntary |
| Norwegian Importer | No obligation | Voluntary |
| Norwegian Distributer | No obligation | Eudamed not open for distributers |
| Foreign Manufacturer | No obligation | Voluntary |
| Foreign Authorised representative | No obligation | Voluntary |
| Foreign Importer | No obligation | Voluntary |
| Foreign Distributer | No obligation | Eudamed not open for distributers |
Registration should be done in the national device register.
Eudamed is the new European database for medical devices, and its use is not mandatory yet.
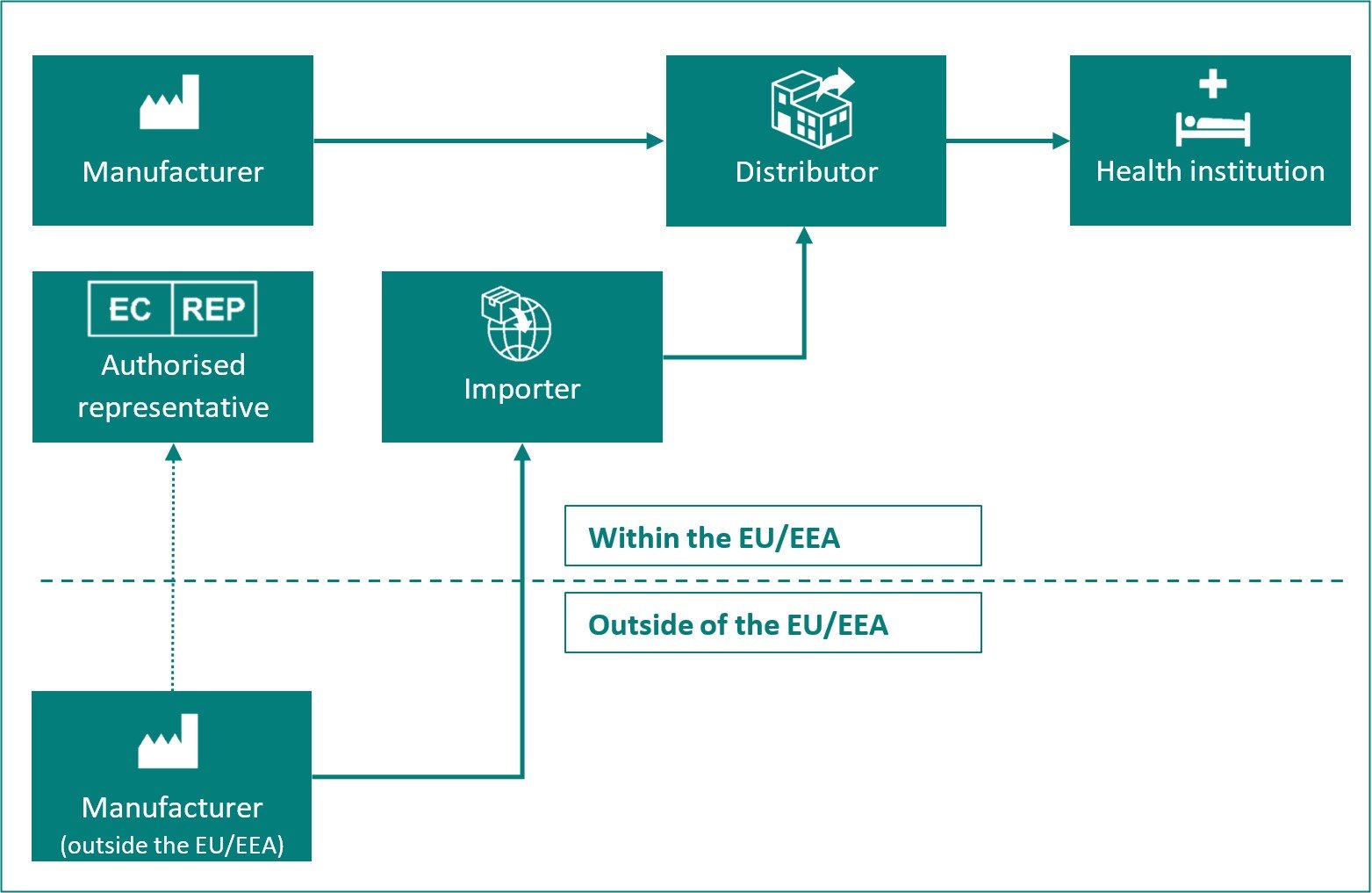
Overview of the different roles of economic operators:
| Economic operator |
Definition in MDR Article 2 and IVDR Article 2 |
| Manufacturer | A natural or legal person who manufactures or fully refurbishes a device or has a device designed, manufactured or fully refurbished, and markets that device under its name or trademark. |
| Authorised representative | Any natural or legal person established within the Union* who has received and accepted a written mandate from a manufacturer, located outside the Union*, to act on the manufacturer's behalf in relation to specified tasks with regard to the latter's obligations under this Regulation. |
| Importer | Any natural or legal person established within the Union* that places a device from a third country on the Union* market. |
| Distributor | Any natural or legal person in the supply chain, other than the manufacturer or the importer, that makes a device available on the market, up until the point of putting into service. |
| Health institution | An organisation the primary purpose of which is the care or treatment of patients or the promotion of public health. |