Application for Substantial Modifications in Clinical Trials of Medicinal Products for Human Use
Published:
Changes
Page contents
What is a Substantial Modification?
After a clinical trial has been approved under the Clinical Trials Regulation (CTR), sponsors may make changes to the trial documentation. These changes are categorised as follows:
- Substantial Modifications (CTR Article 2.2.13): Applications for substantial modifications must be submitted in the Clinical Trials Information System (CTIS).
- Non-Substantial but Relevant Changes (CTR Article 81.9): Sponsors must update relevant information in CTIS for changes that are not substantial but are relevant for Member States to monitor the trial.
- Non-Substantial Changes: Changes that are neither substantial nor relevant for Member States’ monitoring do not require submission in CTIS.
A change is considered substantial if:
- It is likely to have a significant impact on the safety, health, or rights of trial participants.
- It is likely to have a significant impact on the reliability or robustness of the data generated during the trial.
Definition of a Substantial Modification (CTR):
"Any change to any aspect of the clinical trial which is made after notification of a decision referred to in Articles 8, 14, 19, 20 or 23 of the CTR and which is likely to have a substantial impact on the safety or rights of the subjects or on the reliability and robustness of the data generated in the clinical trial."
It is the sponsor’s responsibility to assess whether a change is substantial. Examples of substantial and non-substantial modifications can be found in the Regulation (EU) No 536/2014 Questions & Answers, Annex IV.
The Application Process
For guidance on how to create the application in CTIS, refer to the CTIS Sponsor Handbook, Chapter 4.3: Create and submit a Substantial Modification application (SM).
An application for a substantial modification may cover:
- Part I,
- Part II, or
- Both Part I and Part II.
Technical Limitations in CTIS: CTIS allows only one application to be processed at a time. This means that an application for a substantial modification to Part I cannot be submitted while another Part I application is still under review.
The authorisation procedure for substantial modifications is described in CTR, Chapter III: Authorisation Procedure for a Substantial Modification of a Clinical Trial.
A checklist of required documentation to be attached to the application and submitted via CTIS can be found in CTR, Annex II: Application Dossier for Substantial Modification.
Cover Letter and Description of Changes
When submitting an application for a substantial modification, a cover letter and a description of the changes (modification description) must be included.
For details on what information should be included in the cover letter and change description, refer to CTR, Annex II.
Sponsors are encouraged to use the templates for the Cover Letter and Modification Description, which can be downloaded from the Clinical Trials Coordination Group (CTCG) website, under the Key Documents section.
If the modification involves changes to structured data originally submitted in CTIS, these must be updated as part of the application.
Documentation
Modified documents or excerpts must be submitted with
- An updated version number and date.
- Two versions of each document:
- A version showing tracked changes between the previous and updated text.
- A clean version showing only the updated text.
If the changes in a document are extensive and widespread, a summary of the changes must be included.
All modified documents must be updated with the correct date and version number.
If the application for a substantial modification involves changes to the information in the CTIS form, the form must also be updated at the time of submission.
If applicable, the application should be supplemented with:
- A data summary,
- An updated benefit-risk assessment, and
- Information on the impact of the changes on participants already enrolled in the trial.
Refer to Regulation (EU) No 536/2014, Annex II, Part E: Supporting Information for further details.
Timelines for a Substantial Modification
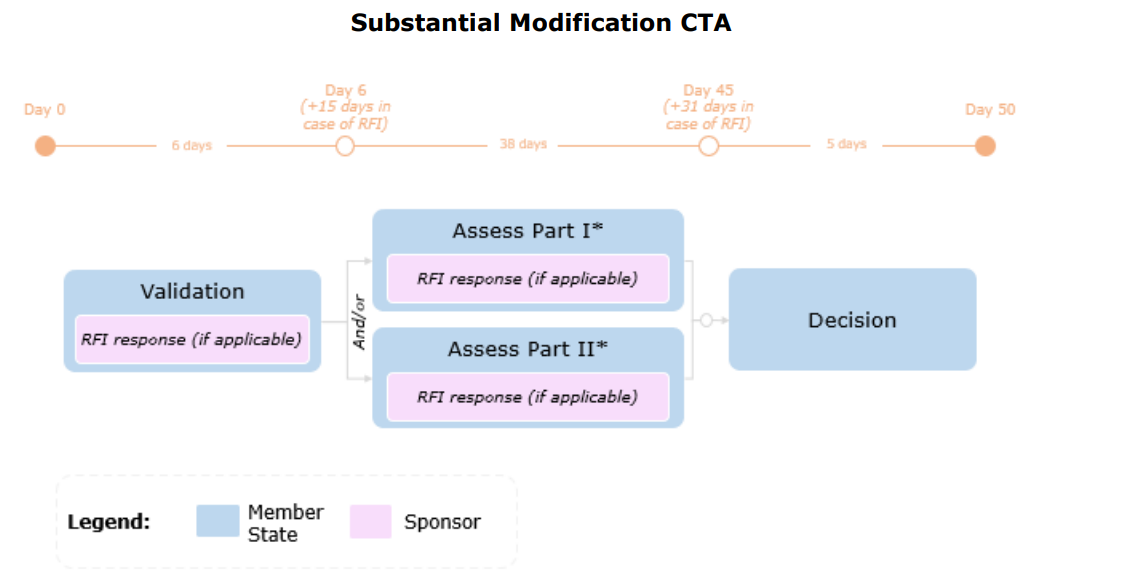
Changes in Accordance with Article 81.9
Structured data in CTIS and/or relevant documents must be updated by the sponsor if changes are made that are not substantial modifications but are relevant for the Member States’ supervision of the clinical trial.
CTR Article 81.9:
"The sponsor shall permanently update in the EU database information on any changes to the clinical trials which are not substantial modifications but are relevant for the supervision of the clinical trial by the Member States concerned."
Non-Substantial Modifications
Non-substantial modifications are those that are neither substantial nor relevant for the Member States’ ability to supervise the clinical trial. These changes do not require submission in CTIS.
Overview in Cover Letter
The cover letter for the next substantial modification application must include an overview of all non-substantial changes made since the last approval.
Documentation
Non-substantial changes must be continuously documented in the Trial Master File.
For more information on non-substantial modifications, refer to Regulation (EU) No 536/2014 Questions & Answers, Section 3.4.
Fees
Invoice information must be provided in Part I (Proof of Payment) of the application in CTIS. An invoice will be issued after the application is received. The invoice will include the EU CT number and DMP reference number. Click here for information on fees for clinical trials of medicinal products.